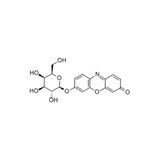
Resorufin ß-D-Galactopyranoside - 25 mg
- Cat.Number : AS-85617
- Manufacturer Ref. :
-
Availability :
In stock
- Shipping conditions : Ice fees will apply
Alternative choices
Unlike FDG that requires a two-step hydrolysis to generate maximum fluorescence, resorufin β-D-galactopyranoside requires only a single-step hydrolysis reaction to attain full fluorescence. This substrate is especially useful for sensitive enzyme measurements in ELISAs.
Specifications
| Chemistry | |
| CAS registry number |
|
|---|---|
| Molecular Formula |
|
| Molecular Mass/ Weight |
|
| Properties | |
| Absorbance (nm) |
|
| Emission (nm) |
|
| Color | |
| Quantity & Purity | |
| Purity |
|
| Storage & stability | |
| Form |
|
| Resuspension condition |
|
| Storage Conditions |
|
| Activity | |
| Application | |
| Biomarker Target | |
| Detection Method | |
| Research Area | |
| Sub-category Research Area | |
| Usage |
|
You may also be interested in the following product(s)

SensoLyte® FDG ß-Galactosidase Assay Kit Fluorimetric - 1 kit

SensoLyte® MUG ß-Galactosidase Assay Kit Fluorimetric - 1 kit

SensoLyte® ONPG ß-Galactosidase Assay Kit Colorimetric - 1 kit
References
A fluorimetric method using fluorescein di-β-d-galactopyranoside for quantifying the senescence-associated β-galactosidase activity in human foreskin fibroblast Hs68 cells.
Anal Biochem . 2004 Feb 15 ; 325(2) 337 | DOI : https://doi.org/10.1016/j.ab.2003.11.012
- NC. Yang
- ML. Hu
Detection of β-galactofuranosidase production by Penicillium and Aspergillus species using 4-nitrophenyl β-D-galactofuranoside
J Appl Bacterio . 1989 Apr 01 ; 66(4) 311 | DOI : https://doi.org/10.1111/j.1365-2672.1989.tb02484.x
- MA. Cousin
- et al
Enzymatic synthesis of p-nitrophenyl 4(5)-O-beta-D-galactosyl-alpha-maltopentaoside as a substrate for human alpha-amylases
Anal Biochem . 1992 Apr 01 ; 202(1) 61 | DOI : https://doi.org/10.1016/0003-2697(92)90206-M
- T. Usui
- et al
Hydrolase activities in brain of neurological mutants: cerebroside galactosidase, nitrophenyl galactoside hydrolase, nitrophenyl glucoside hydrolase and sulphatase
JNC . 1969 Mar 01 ; 16(3) 457 | DOI : 10.1111/j.1471-4159.1969.tb10386.x
- DM, Bowen
- NS. Radin
Hydrolytic activity of alpha-galactosidases against deoxy derivatives of p-nitrophenyl alpha-D-galactopyranoside
Carbohydr Res . 2000 Feb 11 ; 324(2) 107 | DOI : https://doi.org/10.1016/S0008-6215(99)00281-5
- W. Hakamata
- et al
In Situ Histochemical Detection of β-Galactosidase Activity in Lung: Assessment of X-Gal Reagent in Distinguishing lacZ Gene Expression and Endogenous β-Galactosidase Activity
Hum Gene Ther . 1997 Sep 01 ; 8(13) 1545 | DOI : https://doi.org/10.1089/hum.1997.8.13-1545
- DJ. Weiss
- et al
Use of beta-maltosides (p-nitrophenyl-beta-D-maltoside, 2-chloro-4- nitrophenyl-beta-D-maltoside and 4-methylumbelliferyl-beta-D-maltoside) as substrates for the assay of neutral alpha-glucosidase from human kidney and urine
Clin Chim Acta . 1996 Jan 31 ; 244(2) 145 | DOI : https://doi.org/10.1016/0009-8981(95)06189-4
- IS. Lukomskaya
- et al
Synthesis of 4-nitrophenyl b-D-fucofuranoside and b-D-fucofuranosyl-(13)-D-mannopyranose: modified substrates for studies on catalytic requirements of b-D-galactofuranosidase
Carbohydr Res . 2000 Jan 12 ; 323(1-4) 7 | DOI : Chiocconi, A. et al.
- A. Chiocconi
- et al